Viruses. Tiny disease vectors known to cause a wide array of diseases ranging from the common cold to Ebola hemorrhagic fever. They have the ability to enter an organism, inject living cells with their own genetic material, and like microscopic cellular hijackers use the very biological processes that keep cells alive to reproduce. But what if these organisms’ innate ability to bypass the immune system could be harnessed? Could viruses be used to directly target disease?
That’s the question TMU’s Julia Liu began asking when she came across an article describing unusual outcomes in a certain group of cancer patients. According to the article, when some cancer patients were infected with measles, the virus somehow led to cancer cells dying, and the patients made miraculous recoveries.
“A very tough challenge…”
“She took on a very tough challenge…” says Liu’s PhD supervisor Dr. Liang-Tzung Lin, who heads TMU’s Molecular Virology and Oncolytics laboratory. For Liu to engineer the virus to target cancerous cells while harnessing natural immune defenses, she’d need to develop a deep understanding of virology, cancer, cancer immunology, viral immunotherapy, and cancer therapy all at the same time, and she’d need to do it within an ambitious timeframe.
As a bright up-and-coming med student, Liu’s interest in research led her to pursue a combined MD/PhD under Dr. Lin’s supervision. Liu built a strong foundation in both medicine and basic research, integrating clinical and research skills that are, Liu says, “critical for the field to advance”.
Dr. Lin’s lab had been cooperating with Dalhousie University in Canada, and this opened the door for Liu to take advantage of TMU’s dual degree program. This international program would give Liu access to expertise and equipment at both labs, and PhD degrees from both institutions. The challenge would be getting everything done within the time frame. After graduating from the 6-year medical program at TMU (with the last two years overlapping with her PhD studies), Liu moved to Nova Scotia. On top of adjusting to a new lab, new culture, and new country, she’d have just two additional years to complete her final series of experiments and the dual doctoral degree program.

Independent and Expressive
The transition “made me more independent and expressive” says Liu, who credits TMU’s technical and lab skill training with making the adjustment to a maritime provincial lifestyle easier. In Canada, Liu would participate in a more active classroom role, while distance from Taipei’s bustling big-city life provided an opportunity to quietly plan experiments. For coordinating technical and planning issues between labs, as well as navigating the two institutions’ administrative environments, communication would be key.
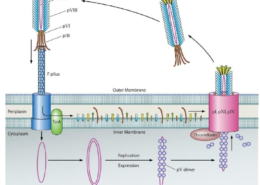
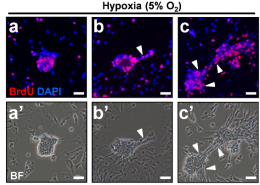
Liu’s research plan was to harness the measles’ oncolytic nature through bioengineering. In effect she wanted to deal cancer cells a one-two punch; the modified measles virus would attack cancerous cells and kill them directly, while the immune system would recognize and destroy targeted cells. “It’s like starting a fire,” says Dr. Lin. “The immune system is like a firefighter who comes in and, while putting out the fire, recognizes that there’s a cancer cell, memorizes it, and triggers an immunity to target the cancer.”
However, breaking down the complex interplay between virus-host-immune-system, Liu would need to work out each step from viral engineering to in-vitro modeling to treatment in animal models. Indeed, the plan was ambitious to the point her supervisors questioned whether she could finish on time. Liu’s schedule was already completely booked, and then COVID-19 hit. Now any mistake could mean waiting a month or more for materials. “We had our fingers crossed the whole time,” says Dr. Lin, who was effectively on call round-the-clock providing technical and moral support from halfway around the world.
A shot against cancer
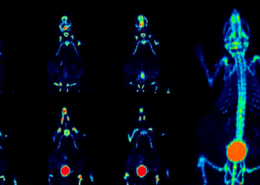
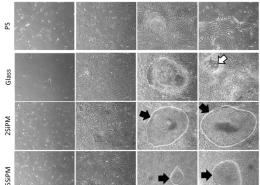
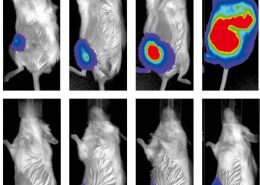
The team organized, and persevered. Liu and Lin’s viral treatment using bioengineered measles infection and immune targeting of virus-marked cells was a resounding double-success. Breast cancer in mice was destroyed, but perhaps even more impressive was the treatment’s activation of the immune system’s “memory”. When Liu reinjected previously treated mice with tumor cells, their immune response immediately went into action, rejecting the tumors outright. If this natural immune system memory persists, relapse could be a thing of the past.
“It’s exciting, but still needs lots of work.” Hurdles remain before bringing the treatment to the public, says Dr. Lin, including confirming long-term safety and developing large-scale production technologies. His lab is now looking at viral bioengineering to target other cancers, or even groups of cancers, to provide both highly specific and broad-spectrum cancer treatments. In the meantime, Dr. Liu is completing her clinical residency while developing therapeutic products with Dr. Lin.
No small feat
With commitment, dedication, and the full support of supervisors halfway around the world, Dr. Liu completed what may have been TMU’s toughest dual degree to date. Thanks to her work, exciting new treatments for cancer are on the horizon, yet Dr. Liu remains humble and soft-spoken. What was it like finishing an MD, a Dual PhD, and harnessing one of nature’s most insidious organisms in the fight against cancer? “It was a great experience overall,” she says.






















-260x185.jpg)















































與連江縣衛生福利局陳美金局長簽署醫療合作備忘錄-260x185.jpg)















期許永續發展成為醫療產業新契機。-260x185.jpg)